| Immunogen | This EGLN1/PHD2 antibody was developed against a synthetic peptide made to an internal portion of mouse PHD2/HIF Prolyl Hydroxylase 2 (between residues 300-400). [Uniprot: Q91YE3] |
| Isotype | IgG |
| Clonality | Polyclonal |
| Host | Rabbit |
| Gene | EGLN1 |
| Purity | Immunogen affinity purified |
| Innovator's Reward | Test in a species/application not listed above to receive a full credit towards a future purchase. |
| Dilutions |
|
||
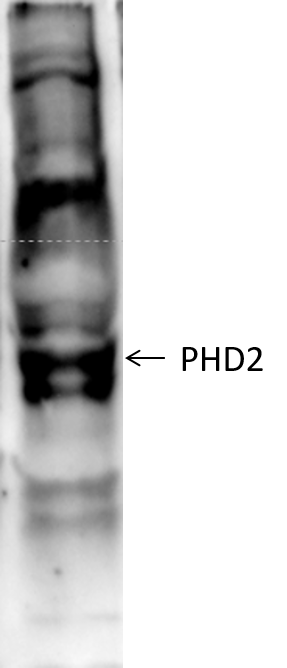
| Application Notes | In Western blot a band is seen ~43 kDa representing HIF Prolyl Hydroxylase 2. There is also a non-specific band of similar intensity at ~75 kDa. |
||
| Theoretical MW | 43 kDa. Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors. |
||
| Control |
|
||
| Control Peptide |
|
||
| Reviewed Applications |
|
||
| Publications |
|
| Storage | Store at 4C. Do not freeze. |
| Buffer | PBS |
| Preservative | 0.02% Sodium Azide |
| Concentration | 1 mg/ml |
| Purity | Immunogen affinity purified |
| Images | Ratings | Applications | Species | Date | Details | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Enlarge |
reviewed by:
Verified Customer |
WB | Mouse | 02/16/2017 |
Summary
|
||||||

Enlarge |
reviewed by:
Helder Andre |
IF | Human | 05/14/2015 |
Summary
|
||||||
|
reviewed by:
Verified Customer |
WB | Mouse | 01/18/2012 |
Summary
|
Secondary Antibodies |
Isotype Controls |
Research Areas for EGLN1/PHD2 Antibody (NB100-2219)Find related products by research area.
|
The concentration calculator allows you to quickly calculate the volume, mass or concentration of your vial. Simply enter your mass, volume, or concentration values for your reagent and the calculator will determine the rest.
5 | |
4 | |
3 | |
2 | |
1 |
| Verified Customer 02/16/2017 |
||
| Application: | WB | |
| Species: | Mouse |
| Helder Andre 05/14/2015 |
||
| Application: | IF | |
| Species: | Human |
| Verified Customer 01/18/2012 |
||
| Application: | WB | |
| Species: | Mouse |
| Gene Symbol | EGLN1 |